Routine surgery can spread cancer women didn't know they had. A doctor in Nazareth found a way to stop it.
The FDA warned about this risk in 2014. A doctor in Nazareth built the answer. Most hospitals still haven't caught up.
She was a doctor. An anesthesiologist at one of the most prestigious hospitals in America. A mother of six. The kind of person who understood medicine better than most patients ever could. And in October 2013, she went in for a routine procedure to remove what her doctors believed were benign uterine fibroids.
A week later, she got a call that changed everything.
The pathology came back. She had leiomyosarcoma, an aggressive form of uterine cancer that looks identical to a fibroid before surgery. There’s no reliable way to definitively diagnose cancer before surgery. Her surgeon had used a power morcellator, a device that breaks tissue into small pieces for removal through a tiny incision, not knowing the fibroid was cancer. The morcellator fragmented the tissue, spreading cancer cells through her body. What would have been a dangerous but treatable stage 1 disease became stage 4. Her five-year survival rate dropped from 50% to somewhere between zero and 20%, according to her husband, Dr. Hooman Noorchashm.
Her name was Dr. Amy Reed. She spent the next four years fighting her cancer, fighting the FDA, fighting the medical establishment that told her this was a freak occurrence. She testified before Congress. She co-authored papers. She went on television, her children in her lap, her head bald from chemotherapy, and she refused to stop talking.
She died in May 2017. She was 44.
Her fight changed the standard of care. After Amy Reed, the FDA revised its risk estimate for hidden uterine cancer at the time of morcellation from 1 in 10,000 to 1 in 350. Johnson & Johnson voluntarily withdrew its morcellators from the market. Hospitals responded.
And yet the problem didn’t go away. It just changed form. Because while hospitals were responding to Amy Reed, they weren’t solving the underlying problem. They were just changing how they created it.
Twelve Years. Same Problem.
OK, I know this sounds complex, so let me break it down.
Every year, more than 500,000 women in the United States alone have surgery to remove their uterus or fibroids. Millions more have similar procedures around the world. In many of these surgeries, the tissue being removed is too large to take out whole through a small incision. So surgeons break it into smaller pieces first, a process called morcellation, and remove it that way.
Here’s the problem. There’s no reliable way to know before surgery whether the tissue contains hidden cancer, and if it does, breaking it apart can spread those cancer cells through the body. With gynecologic cancers on the rise, the stakes of getting this right are only getting higher.
And it’s not just cancer. The same process can spread endometriosis and other tissue-based conditions, affecting women far beyond those with cancer, causing years of chronic pain and complications that could have been prevented.
After the FDA’s 2014 warning, many surgeons stopped breaking tissue apart during surgery and switched to cutting the body open instead, which means a larger incision, longer recovery, more pain, and more risk. Others kept breaking the tissue apart but switched to using ordinary surgical bags never designed for this purpose, bags that according to published studies rupture up to 30% of the time.
Even the better, FDA-cleared containment systems on the market have reported failure rates of around 15% in published data. And a 2025 survey of first-year GYN surgeons in training across US academic hospitals found something troubling: the next generation is being trained right now without proper containment as standard practice. They’re learning the unsafe way from their teachers, and passing it on.
Twelve years after the FDA’s warning, the risk that killed Amy Reed hasn’t been eliminated. It’s been normalized.
As Dr. Vadim Morozov, Professor of Obstetrics and Gynecology at Georgetown University, put it: “We wouldn’t operate with a ruptured glove. So why would we operate with a ruptured containment bag?”
The American medical establishment didn’t have a good answer to that question. A doctor in Nazareth, Israel did.
Israel Is Already in the Room
Here’s what most people don’t know.
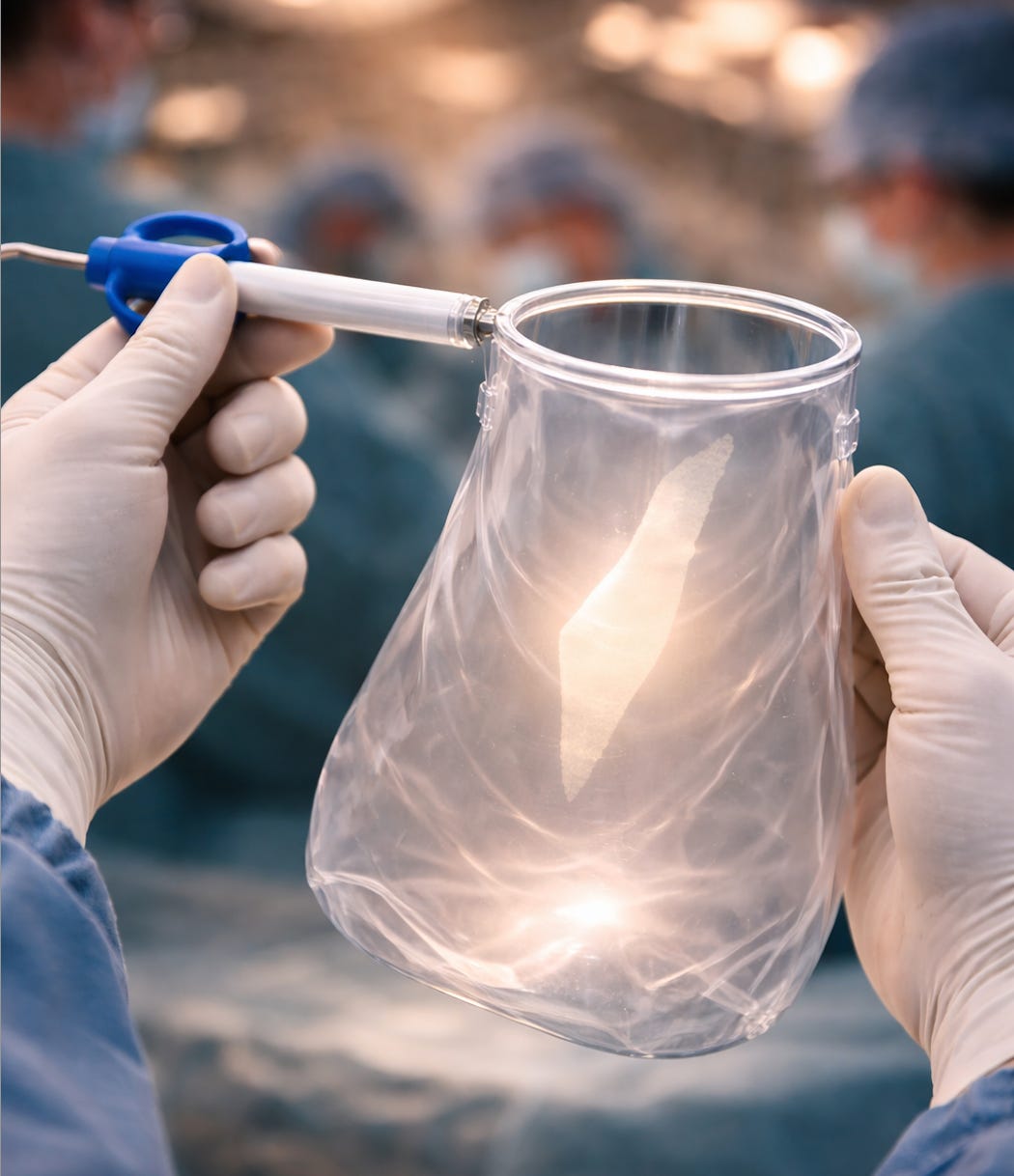
Since November 2023, women in American hospitals have been going into surgery for fibroids and hysterectomies with something new in the operating room. Arizona Gynecology Consultants were among the first to use it. Since then it’s spread to hospitals across the United States. A sealed containment system, purpose-built and FDA-cleared, designed so that whatever the surgeon needs to remove stays completely contained from the first moment of the procedure. If there’s hidden cancer in that tissue, it’s designed to stay contained until it can be properly removed and tested.
It was built in Nazareth, Israel.
Ark Surgical was founded in 2017. Its co-founder and chief scientist, Dr. Abraham Yaari, graduated from Hebrew University Jerusalem, specialized in women’s surgery in New York, and now practices at Saint Mary’s Hospital in Connecticut. As a surgeon who performs these procedures himself, he saw the problem firsthand. The existing containment tools were awkward, slowed surgery down, and had learning curves steep enough that most surgeons simply didn’t bother. So the team built one surgeons would actually use.
The device is called LapBox. Think of it like an airbag for surgery. Just as a car’s airbag deploys around you to absorb impact before you even feel it, LapBox deploys around the tissue before the surgeon begins, creating a sealed environment so that whatever is inside stays inside. It has a dual-wall design with an air-lock that also protects surrounding organs. It works with the way surgeons already operate, including robotic-assisted surgery, which is one of the fastest-growing areas in medicine. The FDA cleared it for one type of surgical tissue removal in 2023, and for a second type in April 2025, making it the first and only FDA-cleared dual-wall containment system for both.
That’s the difference between hoping for the best and having a system designed to protect you.
Dr. Amy Garcia, a GYN surgeon at the University of New Mexico Health Sciences Center, has used it: “The LapBox Tissue Containment System provides the confidence we need to perform the safest and most efficient tissue removal possible.”
If you’re a woman scheduled for fibroid surgery or a hysterectomy, there’s one question worth asking your surgeon before you go in: what containment system do you use, and is it FDA-cleared for morcellation? That question could change your outcome.
None of this happened by accident. Here’s who built it.
Who Decided Enough Was Enough
The chairman of Ark Surgical is Zohar Gendler, a name well known in the Israeli medical device world. He’s led the establishment of more than 50 companies. Mazor Robotics, sold to Medtronic for $1.6 billion. Corindus, sold to Siemens Healthineers for $1.1 billion. Prolor Biotech, sold to OPKO for $480 million. ReWalk, listed on NASDAQ. When Zohar Gendler backs a company, people pay attention.
Stav Tori, the CEO, brings 15 years of medical device experience across some of Israel’s most respected companies, working alongside Chief Medical Officer Dr. Edward Evantash, a former VP of Global Medical Affairs at Hologic, and co-founder Aaron Feldman, the former CEO of Rafael Medical Technologies.
The women building this company are worth naming. Board director Anne Morrissey was the CEO of Alydia Health, acquired for $240 million. Daniela Schardinger, leading marketing and industry strategy, sits on the Innovation Equity Steering Committee of both the Gates Foundation and NIH, and on the World Economic Forum’s Women’s Health Alliance. Livnat Ben-Zur, Director of Business Development, brings over 20 years of medical device and healthcare experience across the US and Israel.
Their advisory board includes the Chair of Obstetrics and Gynecology at Yale School of Medicine, the Head of the Gynecological Department at Sheba Medical Center, and the President of the New European Surgical Academy.
And behind all of it: the Israeli government. Ark Surgical has received grants from the Israel Innovation Authority and the Ministry of Economy and Industry of Israel. This isn’t just a startup that happened to be founded in Israel. It’s a company the Israeli state identified and supported.
Israel Is Already There.
A doctor trained in Jerusalem looked at what the world was accepting and decided it wasn’t good enough.
The world accepted that women going in for routine surgery carried a risk that couldn’t be fully controlled. That the tools were imperfect but close enough. Dr. Yaari and his team built the solution. They earned FDA clearance, twice. And now, in hospitals across America, during the most vulnerable moment of a woman’s medical life, there is an Israeli device in the room making sure she’s protected.
Most of those women will never know where it came from.
We know. And it matters.
To Stav Tori, Dr. Abraham Yaari, Zohar Gendler, Daniela Schardinger, Anne Morrissey, Livnat Ben-Zur, and the entire Ark Surgical team: you are already there, in the room, doing the work nobody else did.
That’s not a small thing. That’s the whole thing. That’s Israel.
Built in Israel. For everyone.
Nothing in this piece is medical advice. If you are facing a gynecologic procedure involving morcellation, speak with your surgeon and ask about containment options.




Wow... thanks for this... I had been contemplating surgery but had no idea of the procedure or the risks... is the device used in Israeli hospitals as well?